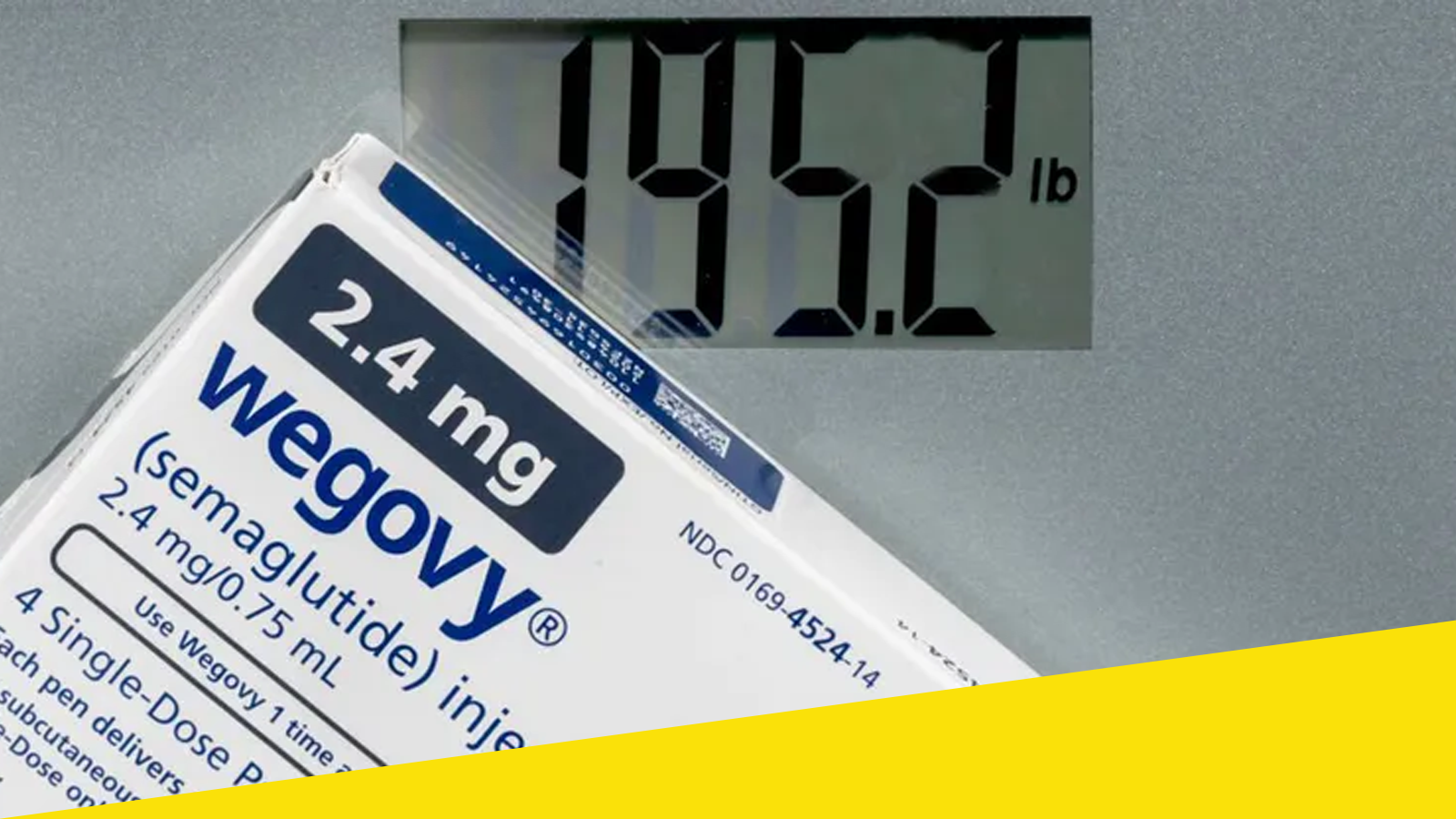
The United States has approved the first pill-based version of Wegovy, marking a major milestone in the treatment of obesity and metabolic disorders. Wegovy, known scientifically as semaglutide, has already transformed weight-loss care through its injectable form, but experts say the new oral option could dramatically expand access and patient adherence.
Obesity affects more than 40 percent of American adults and is closely linked to conditions such as type 2 diabetes, heart disease, and hypertension. While injectable GLP-1 drugs like Wegovy have shown significant success in helping patients lose weight, many individuals have been hesitant to begin treatment due to fear of injections or difficulties with long-term use. The pill version removes this barrier, offering a simpler and more familiar method of treatment.
Clinical trials showed that the oral formulation delivers comparable benefits, helping patients reduce body weight while improving blood sugar control and cardiovascular risk factors. Health professionals believe this approval could normalize medical treatment for obesity, reinforcing the idea that it is a chronic disease rather than a lifestyle failure.
The move is also expected to intensify competition in the booming weight-loss drug market, potentially influencing pricing and availability over time. However, concerns remain about affordability and insurance coverage, as demand for obesity medications continues to outpace supply.
Overall, the approval of a pill-based Wegovy represents a significant step forward in public health. By making effective obesity treatment easier to access and sustain, the decision could reshape how millions of people manage weight and long-term health in the years ahead.